Research Forum
C60, aka fullerene, aka BuckyBalls
Quote from Dr. Garrett Smith on September 6, 2019, 10:09 pmWhat is C60?
Lots of research on C60 here: https://selfhacked.com/blog/fullerene-top-10-benefits-c60-oil/
A great researcher on fullerenes is Aoshima H, see here: https://www.ncbi.nlm.nih.gov/pubmed/?term=%22Aoshima+H%22%5BAuthor%5D
Glyphosate is an organophosphorus compound:
Two organophosphorus compounds glyphosate (Roundup, Vision) {N-(phosphonomethyl)glycine} and glufosinate (Basta) {N-(phosphonomethyl)homoalanine} are broad-spectrum, non-selective herbicides.
Glyphosate Fact Sheet From Pesticides Action Network UK, 1996
In pure chemical terms glyphosate is an organophosphate in that it contains carbon and phosphorous. However, it does not affect the nervous system in the same way as organophosphate insecticides, and is not a cholinesterase inhibitor.
Glyphosate Pesticide Information Profile
Glyphosate is usually formulated as an isopropylamine salt. While it can be described as an organophosphorus compound, glyphosate is not an organophosphate ester but a phosphanoglycine, and it does not inhibit cholinesterase activity.
Reduced negative effects of glyphosate on plants when C60 is present:
Bioactivity Study of the C60-L-Threonine Derivative for Potential Application in Agriculture
The pretreatment of plants with C60-Thr, L-threonine, and nanocomposition containing the fullerene derivative, as well as macro- and microelements, did not have a significant effect on the state of plants in the presence of imidacloprid and carbendazim. At the same time, negative influence of glyphosate herbicide on the grain productivity of plants was reduced by 29%. The leaves of plants treated with C60-Thr containing nanocomposition and glyphosate had a maximal increase of SOD activity with a concomitant decrease of the lipid peroxidation level in comparison with control plants (treated by water and water solution of macro- and microelements with or without glyphosate herbicide treatment; see Table 5). The obtained experimental results of the C60-Thr and C60-Thr derivative influence on the plant antioxidant system can be explained by the immunomodulatory effect of these substances.
[...]
A positive effect of the developed nanocomposition containing C60-Thr on plant resistance to glyphosate is obviously associated with the significant activation of protective plant antioxidant systems.Reduces the effect of organophosphate insecticide effects (glyphosate is an organophosphorus compound, and C60 reduced the effect of glyphosate on plants above):
Fullerene antioxidants decrease organophosphate-induced acetylcholinesterase inhibition in vitro.
Graphene is a flat sheet of carbon atoms arranged in hexagons, while C60 is a spherical arrangement of carbon atoms in hexagons and pentagons. Same carbon, different arrangements of atoms. Graphene put into 3-dimensional shapes (think buckyballs, but a bit different) can aDsorb (detox) glyphosate:
Glyphosate removal from water by functional three-dimensional graphene aerogels
Reduction in muscle fatigue:
C60 fullerene as promising therapeutic agent for correcting and preventing skeletal muscle fatigue
Conclusions - C60FAS leads to reduction in the recovery time of the muscle contraction force and to increase in the time of active muscle functioning before appearance of steady fatigue effects. Therefore, it is possible that C60FAS affects the prooxidant-antioxidant muscle tissue homeostasis, subsequently increasing muscle endurance.
Longevity in rats study:
The prolongation of the lifespan of rats by repeated oral administration of [60]fullerene
Countless studies showed that [60]fullerene (C60) and derivatives could have many potential biomedical applications. However, while several independent research groups showed that C60 has no acute or sub-acute toxicity in various experimental models, more than 25 years after its discovery the in vivo fate and the chronic effects of this fullerene remain unknown. If the potential of C60 and derivatives in the biomedical field have to be fulfilled these issues must be addressed. Here we show that oral administration of C60 dissolved in olive oil (0.8 mg/ml) at reiterated doses (1.7 mg/kg of body weight) to rats not only does not entail chronic toxicity but it almost doubles their lifespan. The effects of C60-olive oil solutions in an experimental model of CCl4 intoxication in rat strongly suggest that the effect on lifespan is mainly due to the attenuation of age-associated increases in oxidative stress. Pharmacokinetic studies show that dissolved C60 is absorbed by the gastro-intestinal tract and eliminated in a few tens of hours. These results of importance in the fields of medicine and toxicology should open the way for the many possible -and waited for- biomedical applications of C60 including cancer therapy, neurodegenerative disorders, and ageing.
Improved wound healing:
A novel class of compounds with cutaneous wound healing properties.
Anti-acne and anti-wrinkle:
Improvement of acne vulgaris by topical fullerene application: unique impact on skin care.
Clinical evaluation of fullerene-C60 dissolved in squalane for anti-wrinkle cosmetics.
Neuroprotective:
Carboxyfullerenes as neuroprotective agents.
Anti-MS:
Reversal of axonal loss and disability in a mouse model of progressive multiple sclerosis
Anti-arthritis:
Inhibition of Inflammatory Arthritis Using Fullerene Nanomaterials
Preventing pre-term birth:
Allergies/asthma:
Anti-tumor:
Tumor-inhibitory effect and immunomodulatory activity of fullerol C60(OH)x.
Increases hair growth:
Fullerene nanomaterials potentiate hair growth.
Applied topically, protects against UV damage of skin:
Medical uses:
Medicinal applications of fullerenes
Protective, anti-inflammatory effects:
Polyhydroxylated C60 fullerene (fullerenol) attenuates neutrophilic lung inflammation in mice.
Fullerene nanomaterials inhibit phorbol myristate acetate-induced inflammation
Carbonic anhydrase inhibition activity (acetazolamide does this, it is a medication used to help reduce symptoms of hypervitaminosis A):
Safety/toxicity:
Sub-acute oral toxicity study with fullerene C60 in rats.
Toxicity studies of fullerenes and derivatives.
Available data clearly shows that pristine C60 has no acute or sub-acute toxicity in a large variety of living organisms, from bacteria and fungal to human leukocytes, and also in drosophila, mice, rats and guinea pigs.
Studies on C60 binding to retinol, forming C60-retinol compounds in the liver (probably making them less reactive until they can be excreted):
In vivo Biology and Toxicology of Fullerenes and Their Derivatives (2008)
Parenteral administration. Intraperitoneal administration of 0.5 mg/kg of a pristine C60 suspension (particle size range: 60–1650 nm) in rats showed that about one-quarter of the dose accumulated in the liver, where the level was approximately constant for 1 week. On day 9 after the administration, the C60 content was about one-third of the plateau level, and on day 13 nearly all C60 was cleared from the liver. The liver contained several metabolites of C60, including its retinol mono-adduct. An intraperitoneal dose of 2 g/kg coloured the liver brown, which had disappeared 2 weeks after the administration. Accumulation was mainly in the Kupffer cells. The macrophages showed a slight hypertrophy without strong activity [38].
In vivo reaction between [60] fullerene and vitamin A in mouse liver (1998)
An in vivo biotransformation of [60] fullerene is observed that does not follow a usual redox metabolic pathway. Following the administration of a single dose of micronized C60 to Swiss mice, C60-retinol and retinyl palmitate adducts were identified in liver by UV/VIS spectroscopy and mass spectrometry after high performance liquid chromatography. NMR investigations of the main biotransformed compound, after in vitro synthesis, show cycloaddition of retinol to the observed biotransformation, which proves that does not remain C60 unchanged in the liver as believed previously, also shows that Diels-Alder-like reactions can occur in vivo.
[60]Fullerene is a Powerful Antioxidant in Vivo with No Acute or Subacute Toxicity (2005)
In the present work, we report the effects of C60-pretreatments on acute carbon tetrachloride intoxication in rats, a classical model for studying free-radical-mediated liver injury. Our results show that aqueous C60 suspensions prepared without using any polar organic solvent not only have no acute or subacute toxicity in rodents but they also protect their livers in a dose-dependent manner against free-radical damage. To be sure, according to histopathological examinations and biological tests, pristine C60 can be considered as a powerful liver-protective agent.
[...]
Analysis of the results obtained for the circulating levels of vitamin E confirmed the protective effect for the lipophilic compartment (Figure 5). Indeed, statistical analysis showed that (i) the median of vitamin E was decreased significantly in the CCl4-treated group in comparison with that of the controls and (ii) there were no differences between the control group and the two groups treated with C60 or with C60 + CCl4.With regard to the circulating levels of vitamin A (Figure 5), statistical analysis showed that there were significant decreases in those groups treated with C60 and/or CCl4 in comparison with that of the control group, whereas there was no difference between the two groups treated with C60. The decrease of vitamin A concentration in the group treated with CCl4 is due to the liver depletion usually observed in the chemically mediated liver fibrosis.17 In the group treated with C60, the decrease of vitamin A concentration may be attributed to a liver depletion consecutive with the reaction with fullerene, leading to either its immobilization and/or to the elimination of the whole adduct after vitamin A-adduct metabolization. These hypotheses must be confirmed by determining the retinol content of the liver as well as by the identification of the “postulated” end products. In the cotreated group (C60 + CCl4), both the postulated mechanisms for the CCl4 and the C60-treated groups could explain the decrease of vitamin A concentration. However, the median in the cotreated group was not decreased significantly in comparison to those of the groups treated with CCl4 and C60. Thus, there was no addition of the effects of C60 and CCl4. This phenomenon is probably due to the protective effect of C60.
[...]
The chromatographic profile of a hepatic extract of a C60-treated rat, obtained under the same conditions as for mouse livers,21 is represented in Figure 2C. This chromatogram shows some additional peaks eluting between those corresponding to the retinyl-esters and the peak of C60, which are absent in hepatic extracts obtained from the control rats. The two main additional peaks correspond to C60-retinol monoadducts formed in the liver following a Diels-Alderlike reaction because (1) they exhibit the same spectral features (Figure 2D) and they have the same retention characteristics as those of the C60-retinol monoadduct we isolated previously from mouse livers;21 and (2) like vitamin A, they are sensitive to air-oxidation and heating, indeed, they can be converted into unmodified C60 after heating at 50 °C for 10 min. These results suggest a possible excretion of C60 via the retinol catabolism. To confirm this new pathway of xenobiotic detoxication, it is necessary for one to isolate and quantify the C60-retinol end products from feces and/or urine.Elimination of [14C(U)]C60 was <2% in urine and feces at any 24 hour time points. [14C(U)]C60 and [14C(U)]C60-retinol were detected in liver of rats and together accounted for ~99% and ~56% of the total recovered at 1 and 30 days post exposure, respectively. The blood radioactivity at 1 hour after [14C(U)]C60 exposure was four-fold higher in rats than in mice; blood radioactivity was still in circulation at 30 days post [14C(U)]C60 exposure in both species (<1%).
[...]
In vivo biotransformation of C60 has been observed in the livers of mice administered micronized C60 via intraperionael injection 7 days after administration. C60-adducts of retinol and of retinyl-palmitate were isolated, providing evidence that C60 in capable of undergoing transformation in vivo by the mouse (Moussa, et al. 1998, Tsuchiya, et al. 1996).
[...]
Extracts of liver collected 1 day and 30 days after exposure to a single tail vein injection of [14C(U)]C60 showed the presence of [14C(U)]C60 and [14C(U)]C60-retinol. The [14C(U)]C60 measured via LC-MS accounted for ~ 99% and ~56% of the radioactivity measured for the liver homogenate by LSC at 1 day and 30 days after exposure, respectively. The chromatographic peak area analysis of [14C(U)]C60 and [14C(U)]C60-retinol indicated the presence of small amounts of [14C(U)]C60-retinol compared to [14C(U)]C60. At 1 day after exposure [14C(U)]C60-retinol peak area was 5.4 ± 0.1% of [14C(U)]C60 total peak area, which decreased to 3.3± 0.8% at 30 days after exposure. [14C(U)]C60-retinol peak area in liver from rats that had five consecutive doses of [14C(U)]C60 was 15% of [14C(U)]C60 total peak area.
[...]
A previous study identified C60-metabolites in the liver of mice by determining the presence of C60-retinol in the liver (Moussa, et al. 1998). Here we confirmed the in vivo formation of a C60-retinol adduct in the liver, detecting [14C(U)]C60-retinol adduct in the liver collected both 1 and 30 day post exposure. However, the 2-fold reduction in the [14C(U)]C60 to [14C(U)]C60-retinol ratio at day 30 after a single tail vein injection compared to 1 day post exposure suggests an overall slow biotransformation of [14C(U)]C60.Poison/"Vitamin A" is mostly stored in the Kupffer cells of the liver,
Hepatic Microanatomy in Hypervitaminosis A in Man and Rat
Hepatic tissue from a patient chronically ingesting excess of Vitamin A and from rats receiving large parenteral doses of Vitamin A palmitate exhibited similar changes. Droplets which stained with oil red 0 and fluoresced under UV transillumination were noted within Kupffer cells in both human and rat specimens. Although the Kupffer cells were greatly enlarged by their content of homogeneous, electron-lucent droplets, the fine structure of cell organelles was intact.
Kupffer cells are also the primary place where nanoparticles like C60 are collected for elimination, which would make them a perfect combo with Poison/"Vitamin A" in those spots:
Kupffer cells are central in the removal of nanoparticles from the organism
The theory at this point is that C60 binds to retinol, reducing its oxidative damage capacity, while readying it for elimination in the liver. If the liver is then able to directly get rid of the c60-retinol complex, without it having to break down further into the more damaging retinoic acids, this would greatly reduce VA-caused inflammation in the system overall.
What is C60?
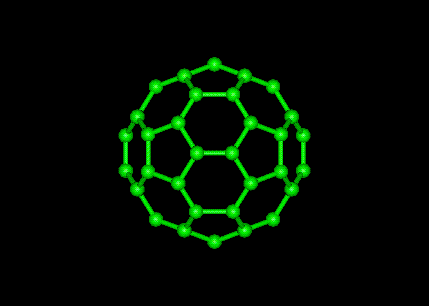
Lots of research on C60 here: https://selfhacked.com/blog/fullerene-top-10-benefits-c60-oil/
A great researcher on fullerenes is Aoshima H, see here: https://www.ncbi.nlm.nih.gov/pubmed/?term=%22Aoshima+H%22%5BAuthor%5D
Glyphosate is an organophosphorus compound:
Two organophosphorus compounds glyphosate (Roundup, Vision) {N-(phosphonomethyl)glycine} and glufosinate (Basta) {N-(phosphonomethyl)homoalanine} are broad-spectrum, non-selective herbicides.
Glyphosate Fact Sheet From Pesticides Action Network UK, 1996
In pure chemical terms glyphosate is an organophosphate in that it contains carbon and phosphorous. However, it does not affect the nervous system in the same way as organophosphate insecticides, and is not a cholinesterase inhibitor.
Glyphosate Pesticide Information Profile
Glyphosate is usually formulated as an isopropylamine salt. While it can be described as an organophosphorus compound, glyphosate is not an organophosphate ester but a phosphanoglycine, and it does not inhibit cholinesterase activity.
Reduced negative effects of glyphosate on plants when C60 is present:
Bioactivity Study of the C60-L-Threonine Derivative for Potential Application in Agriculture
The pretreatment of plants with C60-Thr, L-threonine, and nanocomposition containing the fullerene derivative, as well as macro- and microelements, did not have a significant effect on the state of plants in the presence of imidacloprid and carbendazim. At the same time, negative influence of glyphosate herbicide on the grain productivity of plants was reduced by 29%. The leaves of plants treated with C60-Thr containing nanocomposition and glyphosate had a maximal increase of SOD activity with a concomitant decrease of the lipid peroxidation level in comparison with control plants (treated by water and water solution of macro- and microelements with or without glyphosate herbicide treatment; see Table 5). The obtained experimental results of the C60-Thr and C60-Thr derivative influence on the plant antioxidant system can be explained by the immunomodulatory effect of these substances.
[...]
A positive effect of the developed nanocomposition containing C60-Thr on plant resistance to glyphosate is obviously associated with the significant activation of protective plant antioxidant systems.
Reduces the effect of organophosphate insecticide effects (glyphosate is an organophosphorus compound, and C60 reduced the effect of glyphosate on plants above):
Fullerene antioxidants decrease organophosphate-induced acetylcholinesterase inhibition in vitro.
Graphene is a flat sheet of carbon atoms arranged in hexagons, while C60 is a spherical arrangement of carbon atoms in hexagons and pentagons. Same carbon, different arrangements of atoms. Graphene put into 3-dimensional shapes (think buckyballs, but a bit different) can aDsorb (detox) glyphosate:
Glyphosate removal from water by functional three-dimensional graphene aerogels
Reduction in muscle fatigue:
C60 fullerene as promising therapeutic agent for correcting and preventing skeletal muscle fatigue
Conclusions - C60FAS leads to reduction in the recovery time of the muscle contraction force and to increase in the time of active muscle functioning before appearance of steady fatigue effects. Therefore, it is possible that C60FAS affects the prooxidant-antioxidant muscle tissue homeostasis, subsequently increasing muscle endurance.
Longevity in rats study:
The prolongation of the lifespan of rats by repeated oral administration of [60]fullerene
Countless studies showed that [60]fullerene (C60) and derivatives could have many potential biomedical applications. However, while several independent research groups showed that C60 has no acute or sub-acute toxicity in various experimental models, more than 25 years after its discovery the in vivo fate and the chronic effects of this fullerene remain unknown. If the potential of C60 and derivatives in the biomedical field have to be fulfilled these issues must be addressed. Here we show that oral administration of C60 dissolved in olive oil (0.8 mg/ml) at reiterated doses (1.7 mg/kg of body weight) to rats not only does not entail chronic toxicity but it almost doubles their lifespan. The effects of C60-olive oil solutions in an experimental model of CCl4 intoxication in rat strongly suggest that the effect on lifespan is mainly due to the attenuation of age-associated increases in oxidative stress. Pharmacokinetic studies show that dissolved C60 is absorbed by the gastro-intestinal tract and eliminated in a few tens of hours. These results of importance in the fields of medicine and toxicology should open the way for the many possible -and waited for- biomedical applications of C60 including cancer therapy, neurodegenerative disorders, and ageing.
Improved wound healing:
A novel class of compounds with cutaneous wound healing properties.
Anti-acne and anti-wrinkle:
Improvement of acne vulgaris by topical fullerene application: unique impact on skin care.
Clinical evaluation of fullerene-C60 dissolved in squalane for anti-wrinkle cosmetics.
Neuroprotective:
Carboxyfullerenes as neuroprotective agents.
Anti-MS:
Reversal of axonal loss and disability in a mouse model of progressive multiple sclerosis
Anti-arthritis:
Inhibition of Inflammatory Arthritis Using Fullerene Nanomaterials
Preventing pre-term birth:
Allergies/asthma:
Anti-tumor:
Tumor-inhibitory effect and immunomodulatory activity of fullerol C60(OH)x.
Increases hair growth:
Fullerene nanomaterials potentiate hair growth.
Applied topically, protects against UV damage of skin:
Medical uses:
Medicinal applications of fullerenes
Protective, anti-inflammatory effects:
Polyhydroxylated C60 fullerene (fullerenol) attenuates neutrophilic lung inflammation in mice.
Fullerene nanomaterials inhibit phorbol myristate acetate-induced inflammation
Carbonic anhydrase inhibition activity (acetazolamide does this, it is a medication used to help reduce symptoms of hypervitaminosis A):
Safety/toxicity:
Sub-acute oral toxicity study with fullerene C60 in rats.
Toxicity studies of fullerenes and derivatives.
Available data clearly shows that pristine C60 has no acute or sub-acute toxicity in a large variety of living organisms, from bacteria and fungal to human leukocytes, and also in drosophila, mice, rats and guinea pigs.
Studies on C60 binding to retinol, forming C60-retinol compounds in the liver (probably making them less reactive until they can be excreted):
In vivo Biology and Toxicology of Fullerenes and Their Derivatives (2008)
Parenteral administration. Intraperitoneal administration of 0.5 mg/kg of a pristine C60 suspension (particle size range: 60–1650 nm) in rats showed that about one-quarter of the dose accumulated in the liver, where the level was approximately constant for 1 week. On day 9 after the administration, the C60 content was about one-third of the plateau level, and on day 13 nearly all C60 was cleared from the liver. The liver contained several metabolites of C60, including its retinol mono-adduct. An intraperitoneal dose of 2 g/kg coloured the liver brown, which had disappeared 2 weeks after the administration. Accumulation was mainly in the Kupffer cells. The macrophages showed a slight hypertrophy without strong activity [38].
In vivo reaction between [60] fullerene and vitamin A in mouse liver (1998)
An in vivo biotransformation of [60] fullerene is observed that does not follow a usual redox metabolic pathway. Following the administration of a single dose of micronized C60 to Swiss mice, C60-retinol and retinyl palmitate adducts were identified in liver by UV/VIS spectroscopy and mass spectrometry after high performance liquid chromatography. NMR investigations of the main biotransformed compound, after in vitro synthesis, show cycloaddition of retinol to the observed biotransformation, which proves that does not remain C60 unchanged in the liver as believed previously, also shows that Diels-Alder-like reactions can occur in vivo.
[60]Fullerene is a Powerful Antioxidant in Vivo with No Acute or Subacute Toxicity (2005)
In the present work, we report the effects of C60-pretreatments on acute carbon tetrachloride intoxication in rats, a classical model for studying free-radical-mediated liver injury. Our results show that aqueous C60 suspensions prepared without using any polar organic solvent not only have no acute or subacute toxicity in rodents but they also protect their livers in a dose-dependent manner against free-radical damage. To be sure, according to histopathological examinations and biological tests, pristine C60 can be considered as a powerful liver-protective agent.
[...]
Analysis of the results obtained for the circulating levels of vitamin E confirmed the protective effect for the lipophilic compartment (Figure 5). Indeed, statistical analysis showed that (i) the median of vitamin E was decreased significantly in the CCl4-treated group in comparison with that of the controls and (ii) there were no differences between the control group and the two groups treated with C60 or with C60 + CCl4.With regard to the circulating levels of vitamin A (Figure 5), statistical analysis showed that there were significant decreases in those groups treated with C60 and/or CCl4 in comparison with that of the control group, whereas there was no difference between the two groups treated with C60. The decrease of vitamin A concentration in the group treated with CCl4 is due to the liver depletion usually observed in the chemically mediated liver fibrosis.17 In the group treated with C60, the decrease of vitamin A concentration may be attributed to a liver depletion consecutive with the reaction with fullerene, leading to either its immobilization and/or to the elimination of the whole adduct after vitamin A-adduct metabolization. These hypotheses must be confirmed by determining the retinol content of the liver as well as by the identification of the “postulated” end products. In the cotreated group (C60 + CCl4), both the postulated mechanisms for the CCl4 and the C60-treated groups could explain the decrease of vitamin A concentration. However, the median in the cotreated group was not decreased significantly in comparison to those of the groups treated with CCl4 and C60. Thus, there was no addition of the effects of C60 and CCl4. This phenomenon is probably due to the protective effect of C60.
[...]
The chromatographic profile of a hepatic extract of a C60-treated rat, obtained under the same conditions as for mouse livers,21 is represented in Figure 2C. This chromatogram shows some additional peaks eluting between those corresponding to the retinyl-esters and the peak of C60, which are absent in hepatic extracts obtained from the control rats. The two main additional peaks correspond to C60-retinol monoadducts formed in the liver following a Diels-Alderlike reaction because (1) they exhibit the same spectral features (Figure 2D) and they have the same retention characteristics as those of the C60-retinol monoadduct we isolated previously from mouse livers;21 and (2) like vitamin A, they are sensitive to air-oxidation and heating, indeed, they can be converted into unmodified C60 after heating at 50 °C for 10 min. These results suggest a possible excretion of C60 via the retinol catabolism. To confirm this new pathway of xenobiotic detoxication, it is necessary for one to isolate and quantify the C60-retinol end products from feces and/or urine.
Elimination of [14C(U)]C60 was <2% in urine and feces at any 24 hour time points. [14C(U)]C60 and [14C(U)]C60-retinol were detected in liver of rats and together accounted for ~99% and ~56% of the total recovered at 1 and 30 days post exposure, respectively. The blood radioactivity at 1 hour after [14C(U)]C60 exposure was four-fold higher in rats than in mice; blood radioactivity was still in circulation at 30 days post [14C(U)]C60 exposure in both species (<1%).
[...]
In vivo biotransformation of C60 has been observed in the livers of mice administered micronized C60 via intraperionael injection 7 days after administration. C60-adducts of retinol and of retinyl-palmitate were isolated, providing evidence that C60 in capable of undergoing transformation in vivo by the mouse (Moussa, et al. 1998, Tsuchiya, et al. 1996).
[...]
Extracts of liver collected 1 day and 30 days after exposure to a single tail vein injection of [14C(U)]C60 showed the presence of [14C(U)]C60 and [14C(U)]C60-retinol. The [14C(U)]C60 measured via LC-MS accounted for ~ 99% and ~56% of the radioactivity measured for the liver homogenate by LSC at 1 day and 30 days after exposure, respectively. The chromatographic peak area analysis of [14C(U)]C60 and [14C(U)]C60-retinol indicated the presence of small amounts of [14C(U)]C60-retinol compared to [14C(U)]C60. At 1 day after exposure [14C(U)]C60-retinol peak area was 5.4 ± 0.1% of [14C(U)]C60 total peak area, which decreased to 3.3± 0.8% at 30 days after exposure. [14C(U)]C60-retinol peak area in liver from rats that had five consecutive doses of [14C(U)]C60 was 15% of [14C(U)]C60 total peak area.
[...]
A previous study identified C60-metabolites in the liver of mice by determining the presence of C60-retinol in the liver (Moussa, et al. 1998). Here we confirmed the in vivo formation of a C60-retinol adduct in the liver, detecting [14C(U)]C60-retinol adduct in the liver collected both 1 and 30 day post exposure. However, the 2-fold reduction in the [14C(U)]C60 to [14C(U)]C60-retinol ratio at day 30 after a single tail vein injection compared to 1 day post exposure suggests an overall slow biotransformation of [14C(U)]C60.
Poison/"Vitamin A" is mostly stored in the Kupffer cells of the liver,
Hepatic Microanatomy in Hypervitaminosis A in Man and Rat
Hepatic tissue from a patient chronically ingesting excess of Vitamin A and from rats receiving large parenteral doses of Vitamin A palmitate exhibited similar changes. Droplets which stained with oil red 0 and fluoresced under UV transillumination were noted within Kupffer cells in both human and rat specimens. Although the Kupffer cells were greatly enlarged by their content of homogeneous, electron-lucent droplets, the fine structure of cell organelles was intact.
Kupffer cells are also the primary place where nanoparticles like C60 are collected for elimination, which would make them a perfect combo with Poison/"Vitamin A" in those spots:
Kupffer cells are central in the removal of nanoparticles from the organism
The theory at this point is that C60 binds to retinol, reducing its oxidative damage capacity, while readying it for elimination in the liver. If the liver is then able to directly get rid of the c60-retinol complex, without it having to break down further into the more damaging retinoic acids, this would greatly reduce VA-caused inflammation in the system overall.
Licensed Naturopathic Physician (NMD) in Arizona
NutritionDetective.com, home of the Love Your Liver program
YouTube - FaceBook - Instagram - Twitter
